By Eldon Muehling – “Dr. Water”. This month we will be look at chlorine and its various properties. We will be emphasizing how chlorine and its by-products can adversely affect our health and what we can do to avoid its harmful effects.
What is chlorine?
Chlorine, with the atomic chemical symbol Cl, is a naturally occurring element with the atomic number of 17 on the periodic chart. A molecule of chlorine is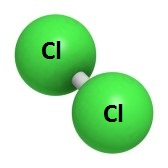 composed of two chlorine atoms with its formula chemically expressed as Cl with a subscript 2. Chlorine, at room temperature, is a pale green, extremely active and poisonous gas with a very distinctive and obnoxious odor. It makes breathing very labored. It is especially dangerous to humans and other animals even in very small quantities as a gas or as a compressed liquid. In its rare solid form, it’s more of a yellow green color. In spite of it serious dangers, it also has many uses which we will look at later, especially for disinfecting and bleaching various substances.
composed of two chlorine atoms with its formula chemically expressed as Cl with a subscript 2. Chlorine, at room temperature, is a pale green, extremely active and poisonous gas with a very distinctive and obnoxious odor. It makes breathing very labored. It is especially dangerous to humans and other animals even in very small quantities as a gas or as a compressed liquid. In its rare solid form, it’s more of a yellow green color. In spite of it serious dangers, it also has many uses which we will look at later, especially for disinfecting and bleaching various substances.
What is chloride?
Chloride on the other hand, is the chemically combined form of chlorine. A chloride ion has the chemical symbol Cl with a single negative charge expressed as super-script -1). This negatively charged particle (ion) is formed whenever the chlorine atom gains one electron from another element, usually a metal. In comparison to the element chlorine, chloride is relatively inert and not nearly as dangerous to our health. It is quite abundant in nature…especially as the compound sodium chloride (NaCl) or common table salt. Humans as well as many other animals need small amounts of chloride (not chlorine) but there is a definite limit as to what amount we can physically tolerate. Chloride is also found naturally in many other salts on earth.
charge expressed as super-script -1). This negatively charged particle (ion) is formed whenever the chlorine atom gains one electron from another element, usually a metal. In comparison to the element chlorine, chloride is relatively inert and not nearly as dangerous to our health. It is quite abundant in nature…especially as the compound sodium chloride (NaCl) or common table salt. Humans as well as many other animals need small amounts of chloride (not chlorine) but there is a definite limit as to what amount we can physically tolerate. Chloride is also found naturally in many other salts on earth.
How am I being exposed to chlorine and its by-products?
There are three common ways that we are exposed to chlorine: first and perhaps the most obvious, by drinking and eating food or water that contains chlorine, secondly, breathing fumes that chlorine creates directly or indirectly when it is combined with other commonly found substances, and thirdly, from direct contact of our skin with water. Even taking a long bath or shower increases a person’s risk for chlorine exposure because chlorine can enter your body through your eyes, nose, even your ears!!
We can even absorb chlorine through skin directly from the air! It should be noted that the dangers of chlorine exposure also exists for those who don’t normally swim, but who instead choose to sit by the pool sunbathing or socializing. Non-swimmers have even tested positive for tri-chloramines, the combined form of chlorine and ammonia which has replaced chlorination in many municipal water supplies. (More on tri-chloramines later) This means that we all need to be more aware of some of the seemingly innocent danger that we are commonly exposed to!
How is and why is chlorine used?
Chlorine has been used to disinfect drinking water for more than 100 years because it does an excellent job of controlling the growth of such unwelcome bacteria as E- coli and other harmful bacteria. Based on earlier successful studies of water disinfection in England, chlorination of water began in Lincoln, England in 1905. Failure of a faulty slow sand filter, which was being used to treat a contaminated water supply, there was an epidemic of typhoid fever. A man by the name of Dr. Alexander Houston fed a concentrated solution of chloride of lime into the water being treated and thus used chlorination to help stop the epidemic. Mainly as a precaution, the chlorination was continued until 1911 when a new water supply was instituted.
coli and other harmful bacteria. Based on earlier successful studies of water disinfection in England, chlorination of water began in Lincoln, England in 1905. Failure of a faulty slow sand filter, which was being used to treat a contaminated water supply, there was an epidemic of typhoid fever. A man by the name of Dr. Alexander Houston fed a concentrated solution of chloride of lime into the water being treated and thus used chlorination to help stop the epidemic. Mainly as a precaution, the chlorination was continued until 1911 when a new water supply was instituted.
The first continuous use of chlorine in the United States for disinfection took place in 1908 at Boonton Reservoir (on the Rockaway River), which served as the supply for Jersey City, New Jersey. The chlorination process, which was first conceived by Dr. John L. Leal, was achieved by the controlled additions of dilute solution of calcium hypochlorite at the rate of 0.2 to 0.35 parts per million. Over the next few years, chlorination was rapidly installed in drinking water systems all around the world.
Are there any substitutes for Chlorine?
Today, by many schools of thought, chlorine has become a dinosaur for swimming pool sanitation. Alternative methods that can be used to keep pools disinfected, including silver‐copper ion generators and ozone systems but they are much more expensive and are subject to other shortcomings and problems. We don’t use chlorine because it is the safest or even the most effective means of disinfection, we use it because it is the cheapest! In spite of all our technological advances, we essentially still pour chlorine bleach (essentially Clorox) into our water before we drink it. What are the symptoms of too much exposure to chlorine?
How Does Chlorine Cause a Health Problem?
Research shows that long‐term exposure to chlorine leads to the production of free radicals within the body. These free radicals are carcinogenic, and cause tremendous damage to our cells. In other words, chlorine causes cancer. Several sources have stated that developing cancer is 93% higher in people who drink or are exposed to chlorine in other ways. Wow. That is almost twice the risk of cancer! Because it is a potential health hazard in both children and adults, this issue should be taken very seriously.
Chlorine reacting with ammonia can also be a health problem.
 As many have learned the hard way, when swimming in a heavily chlorinated swimming pool, chlorine can irritate and burn one’s eyes and throat …it can even irritate or burn skin. This may be the reason why. When chlorine reacts with ammonia, mono- chloramines, di-chloramines or tri-chloramines are formed, depending on the chemical ratio. According to a 2003 Belgian study, these skin irritants are released any time chlorinated water reacts with organic materials such as human sweat or urine. In addition, tri-chloramines are believed to initiate a biological process that effectively destroys the cellular barriers surrounding the lungs. Perhaps this explains why children exposed to large amounts of chlorine often suffer asthmatic attacks. In another research study, it was learned that rats exposed to chlorine and chloramines developed tumors in their kidneys and intestines. And there is more…
As many have learned the hard way, when swimming in a heavily chlorinated swimming pool, chlorine can irritate and burn one’s eyes and throat …it can even irritate or burn skin. This may be the reason why. When chlorine reacts with ammonia, mono- chloramines, di-chloramines or tri-chloramines are formed, depending on the chemical ratio. According to a 2003 Belgian study, these skin irritants are released any time chlorinated water reacts with organic materials such as human sweat or urine. In addition, tri-chloramines are believed to initiate a biological process that effectively destroys the cellular barriers surrounding the lungs. Perhaps this explains why children exposed to large amounts of chlorine often suffer asthmatic attacks. In another research study, it was learned that rats exposed to chlorine and chloramines developed tumors in their kidneys and intestines. And there is more…
Chlorine, Cancer, and Heart Disease
In the late sixties, Dr. Joseph Price wrote a highly controversial book titled, “Coronaries, Cholesterol and Chlorine”. In this book, Dr. Price concluded that chlorine causes fat to grow around the heart and he believed this to be the basic cause of atherosclerosis, heart attacks and stroke.
Dr. Price later headed up a study using chickens as test subjects, where two groups of several hundred birds were observed throughout their span to maturity. One group was given water with chlorine and the other without. The group raised with chlorine, when autopsied, showed some level of heart or circulatory disease in every specimen, the group without had no incidence of disease. The group with chlorine under winter conditions, showed outward signs of poor circulation, shivering, drooped feathers and a reduced level of activity. The group without chlorine grew faster, larger and displayed vigorous health. This study was well received in the poultry industry and is still used as a reference today. As a result, most of the large poultry producers use dechlorinated water. It should be common sense to conclude that if regular use of chlorinated tap water is not good for chickens, then it should not be good for humans!
Chlorine By-Products Cause Cancer
There is another concern about chlorine. When chlorine is added to our water, it often combines with other natural compounds, such as humic acid, to form tri-halo-methanes (by-products of chlorine, fluorine and bromine). The most common of these products is chloroform, a known carcinogen. Although the concentrations of these carcinogens (THMs) are low, it is precisely these low levels that cause cancer, and many scientists believe them to be responsible for the majority of human cancer in the United States!
Good News!
The good news is that chlorine is one of the easiest substances to remove from our water. For that reason it logically should continue to serve its purpose of keeping our water free from harmful bacteria and water-borne disease right up to the time of consumption. Then it should be removed by a quality water treatment device such as a home water distiller in combination with an activated carbon filter. The simple truth is that chlorine is most likely here to stay. The very idea that we can do away with chlorine any time soon is simply not realistic. We should however, recognize the serious threats that chlorine and its by-products are to our health. This means that chlorine and any of its byproducts should be removed in our homes at the point-of-use, both from the water that we drink and the water that we shower or bathe in.
home water distiller in combination with an activated carbon filter. The simple truth is that chlorine is most likely here to stay. The very idea that we can do away with chlorine any time soon is simply not realistic. We should however, recognize the serious threats that chlorine and its by-products are to our health. This means that chlorine and any of its byproducts should be removed in our homes at the point-of-use, both from the water that we drink and the water that we shower or bathe in.
REFERENCES
1. The Hidden Danger of Chlorine in our Bath Water. http://ezinearticles.com/?The‐Hidden‐Danger‐Of‐Chlorine‐
